da Vinci® Pancreatectomy (non-cancerous)
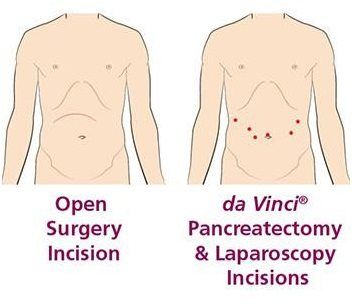
If you are facing a disease that affects your pancreas, your doctor may suggest surgery. Surgery to remove the pancreas, or parts of the pancreas, is called a pancreatectomy. It can be done using open surgery through one large incision or minimally invasive surgery (through a few small incisions) using da Vinci Surgery or traditional laparoscopy.
Why da Vinci Surgery?
da Vinci technology enables your surgeon to operate through a few small incisions (cuts), like traditional laparoscopy, instead of a large open incision. The da Vinci System is a robotic-assisted surgical device that your surgeon is 100% in control of at all times. The da Vinci System gives surgeons:
- A 3D HD view inside your body
- Wristed instruments that bend and rotate far greater than the human hand
- Enhanced vision, precision and control
 The da Vinci System has brought minimally invasive surgery to more than 3 million patients worldwide. da Vinci technology – changing the experience of surgery for people around the world.
The da Vinci System has brought minimally invasive surgery to more than 3 million patients worldwide. da Vinci technology – changing the experience of surgery for people around the world.
- To locate a da Vinci surgeon nearest you, use our surgeon locator
- Learn more about the da Vinci Surgical System.
Risks & Considerations Related to Pancreatic Surgery (removal of all/part of the pancreas): inflamed pancreas, leak of pancreatic or bile juices, narrowing or leaking at the spot where the pancreas is connected to the bowel, injury to the spleen or bowel, insufficient pancreatic function (for example, diabetes), diarrhea, poor stomach emptying.
IMPORTANT SAFETY INFORMATION
Serious complications may occur in any surgery, including da Vinci® Surgery, up to and including death. Individual surgical results may vary. Patients should talk to their doctor to decide if da Vinci Surgery is right for them. Patients and doctors should review all available information on non-surgical and surgical options in order to make an informed decision. Please also refer to http://www.daVinciSurgery.com/Safety for Important Safety Information. ©2017 Intuitive Surgical, Inc. All rights reserved. Product names are trademarks or registered trademarks of their respective holders. The information on this website is intended for a United States audience only. PN 1006497 Rev B 11/15


